 By Jophin February 25, 2026
min read
By Jophin February 25, 2026
min readAI in Drug Discovery: Accelerating Medical Breakthroughs and Reducing Development Time
The pharmaceutical industry faces a critical challenge: developing a single drug cost approximately $2.6 billion and takes 10-15 years, with a 90% failure rate in clinical trials. For C – suite executives navigating this landscape, AI drug discovery represents not just an efficiency upgrade, but a fundamental reimagining of how medicines reach patients.
The Economic Reality Driving AI Adoption in Drug Discovery
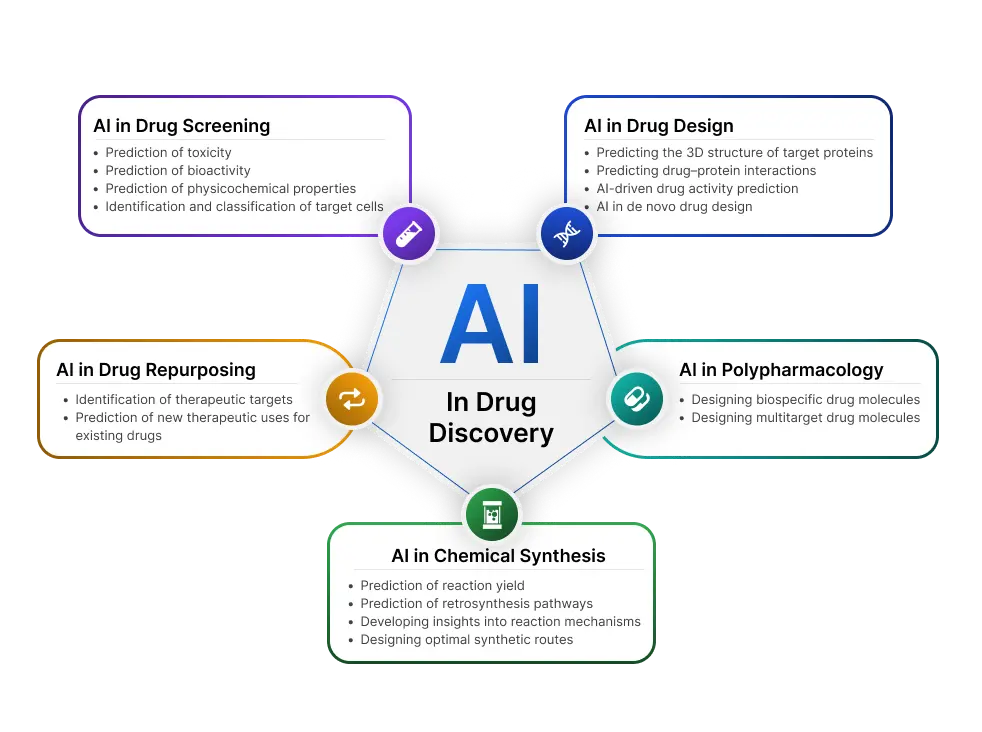
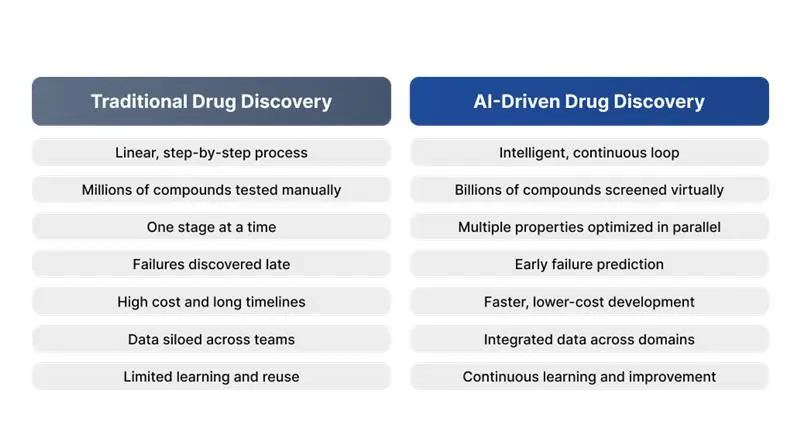
Traditional drug development follows a linear, resource-intensive path. Pharmaceutical companies screen millions of compounds, invest years in preclinical studies, and navigate complex regulatory requirements before achieving market approval. AI in drug discovery compresses these timelines by identifying promising drug candidates faster, predicting molecular behavior with unprecedented accuracy, and reducing costly late – stage failures. These are supported by medical device integration platforms that connect laboratory systems, research databases, and clinical data into a unified foundation for AI analysis.
The financial implications are substantial. Companies implementing AI drug discovery platforms report 40 – 60% reductions in preclinical development time and significant cost savings in compound screening phases. For executives evaluating ROI, these aren’t marginal improvements; they represent competitive advantages that directly impact pipeline velocity and shareholder value.
How AI Transforms Drug Discovery Across Development Stages
Target Identification and Validation
AI algorithms analyze genomic data, protein structures, and disease pathways to identify therapeutic targets that human researchers might overlook. Machine learning models process vast biomedical literature, millions of research papers, clinical trial results, and molecular databases – to uncover connections between diseases and potential drug targets.
AI systems accelerate target validation by predicting how specific proteins interact with disease mechanisms, allowing organizations to fail faster on unpromising targets and concentrate resources on candidates with higher success probability.
Compound Screening and Lead Optimization
Traditional high-throughput screening tests thousands of compounds physically in laboratories, requiring extensive infrastructure and time. AI drug discovery platforms can virtually screen billions of molecular structures, predicting binding affinity, toxicity, and efficacy before synthesizing a single molecule.
Generative AI models now design novel molecular structures optimized for specific therapeutic properties. These systems learn from existing drug databases and propose entirely new compounds that meet multiple criteria simultaneously: target specificity, minimal side effects, favorable pharmacokinetics, and manufacturing feasibility. What previously required extensive medicinal chemistry expertise and iterative laboratory work now begins with computational precision.
The lead optimization phase benefits particularly from AI’s ability to predict how molecular modifications affect multiple characteristics simultaneously. Rather than sequential testing of structural variations, AI models suggest modifications that optimize all relevant parameters.
Clinical Trial Optimization
AI in drug discovery extends beyond laboratory phases into clinical development. Predictive models identify patient populations most likely to respond to treatments, enabling more targeted trial design. Natural language processing extracts insights from electronic health records to inform trial protocols, while machine learning algorithms monitor patient data in real – time to detect safety signals earlier.
For pharmaceutical executives, this translates to faster enrolment, better patient stratification, and more efficient resource allocation across trial sites. Organizations that leverage AI for clinical development gain the ability to make data – driven decisions about trial modifications and endpoint selection during ongoing studies.
Real – World Impact: Beyond the Hype
The pharmaceutical industry has witnessed tangible progress in AI drug discovery moving from research concept to clinical reality. Insilico Medicine demonstrated this transition by identifying both a novel target and designing a drug candidate in 18 months, progressing from target discovery to Phase 1 clinical trials in under 30 months. Their drug candidate for idiopathic pulmonary fibrosis, Rentosertib, has since shown promising results in Phase 2a trials, marking the first time an AI – designed drug for an AI – discovered target has demonstrated clinical efficacy in patients.
Exscientia has collaborated with major pharmaceutical companies to advance multiple AI – designed molecules into clinical development. These examples represent proof points that matter to boards and investors: AI drug discovery delivers measurable outcomes affecting pipeline quality, development timelines, and competitive positioning.
Strategic Implementation Considerations for Leadership
Data Infrastructure Requirements
Successful AI drug discovery depends on high – quality, integrated data. Many pharmaceutical companies operate with siloed datasets across research units and legacy systems that don’t communicate effectively. Executives must prioritize data standardization, ensure interoperability between laboratory systems, and invest in computational infrastructure capable of processing complex molecular simulations.
The strategic question isn’t whether your organization has data; most pharmaceutical companies possess extensive compound libraries and research archives – but whether that data is structured and accessible for AI applications. Organizations that treat data infrastructure as foundational create sustainable advantages as AI capabilities evolve.
Talent and Partnership Models
Building internal AI drug discovery capabilities requires specialized talent: computational chemists, machine learning engineers, and bioinformaticians who bridge pharmaceutical development with AI methodologies. Given talent scarcity, many organizations adopt hybrid models: developing core AI competencies internally while partnering with specialized platforms for specific applications.
This approach allows pharmaceutical companies to maintain proprietary control over critical research while accessing cutting – edge AI capabilities. For CFOs evaluating capital allocation, partnerships often provide faster time – to – value than building comprehensive AI infrastructure from scratch.
Regulatory Considerations and Validation
Regulatory agencies are developing frameworks for AI-driven drug development, but standards remain evolving. Pharmaceutical executives must ensure AI-generated insights are explainable, reproducible, and meet regulatory scrutiny. This means implementing validation protocols that demonstrate how AI systems arrive at predictions and maintaining detailed documentation of AI – assisted decision – making throughout the development process.
The FDA and EMA have issued preliminary guidance on AI in drug development, emphasizing transparency and validation. Organizations that proactively engage regulators and establish robust AI governance frameworks position themselves favorably for future approvals.
Measuring Success: Metrics That Matter
Traditional pharmaceutical metrics, compound progression rates, trial enrollment speed, regulatory timelines – remain relevant, but AI drug discovery introduces additional indicators. Track AI – recommended compounds advancing through stages, prediction of accuracy, and time savings.
Measure portfolio diversification and ability to explore novel hypotheses. AI enables investigation of unconventional targets that might be too resourceful – intensive traditionally.
Market Forces Accelerating Adoption
The combination of patent losses, costly R&D, and shrinking pricing flexibility is driving an urgent need to improve development productivity. Companies that reduce time – to – market gain competitive advantages through extended market exclusivity.
Rare disease treatments and personalized medicine benefit particularly from AI drug discovery, representing expanding opportunities where AI efficiently navigates limited data.
Why Consider Fortunesoft for Your AI Drug Discovery Journey
Implementing AI drug discovery requires transformation of research workflows, data infrastructure, and organizational capabilities. Many pharmaceutical executives recognize AI’s potential but struggle with practical implementation: integrating AI platforms with existing systems, training research teams, ensuring regulatory compliance, and demonstrating ROI.
Fortunesoft has guided pharmaceutical and biotech companies through this transition with strategic implementation support. Our teams are deeply familiar with the realities of pharmaceutical R&D, from fragmented legacy data and strict regulatory demands to developing in-house capabilities while still driving measurable impact.
We help organizations structure data for AI applications, implement validated platforms, and build lasting competitive advantages. This includes enabling downstream clinical development through clinical trial management software that supports AI-driven patient stratification, real-time trial monitoring, and regulatory-ready data governance. Our approach combines technical excellence with change management expertise.
Whether you’re exploring AI drug discovery for the first time or scaling existing initiatives, we focus on incremental value creation: identifying high – impact use cases, building confidence through early wins, and expanding AI capabilities as your team develops expertise.
Future Trajectories and Strategic Positioning
AI drug discovery continues evolving with emerging technologies. Multi-modal AI systems integrating genomics, proteomics, and clinical outcomes will provide increasingly holistic insights.
Organizations establishing AI drug discovery capabilities today position themselves to leverage these advances. For C – suite leaders, AI drug discovery isn’t simply about speed; it’s about fundamentally changing what’s possible in treating diseases that currently lack effective therapies.
FAQs
How long does it take to see ROI from AI drug discovery investments?
Organizations typically observe initial ROI within two years through reduced screening costs and faster lead identification. Full financial impact becomes evident when AI – discovered compounds advance through clinical trials.
What types of diseases benefit most from AI drug discovery?
AI shows particular promise in oncology, neurodegenerative diseases, rare genetic disorders, and infectious diseases – areas where traditional screening struggles with target complexity.
How do pharmaceutical companies protect IP when using AI platforms?
Most platforms keep proprietary data within your infrastructure. Companies maintain ownership of AI – generated insights, discovered compounds, and novel targets through clear contractual agreements.
Can AI replace medicinal chemists and researchers?
AI strengthens and extends human capabilities instead of substituting for them. Successful implementations combine AI’s computational power with researchers’ domain expertise and scientific intuition.
What are the main risks in implementing AI drug discovery?
Primary risks include data quality issues, over-reliance on AI without validation, regulatory uncertainty, and integration challenges. Mitigating these requires phased implementation with validation protocols and ongoing training.
Sources
Author Bio


 Facebook
Facebook Whatsapp
Whatsapp LinkedIn
LinkedIn Pinterest
Pinterest






 Start Chat
Start Chat